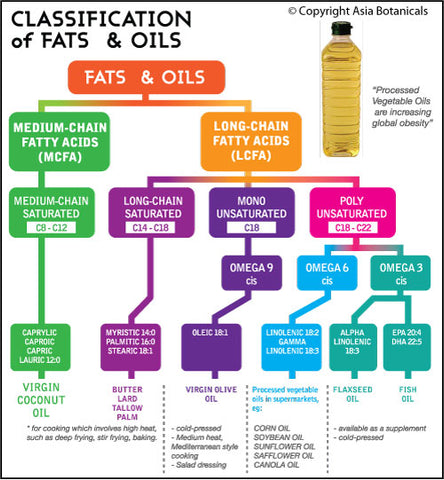
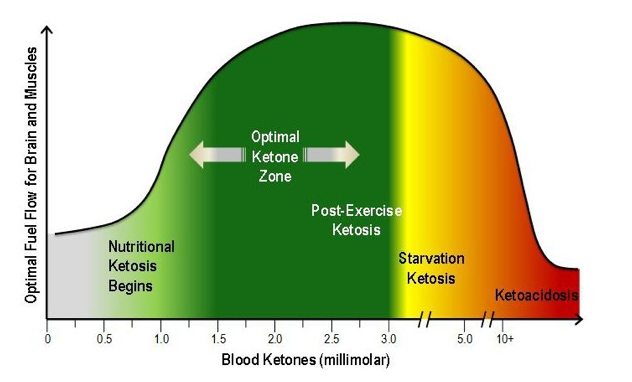
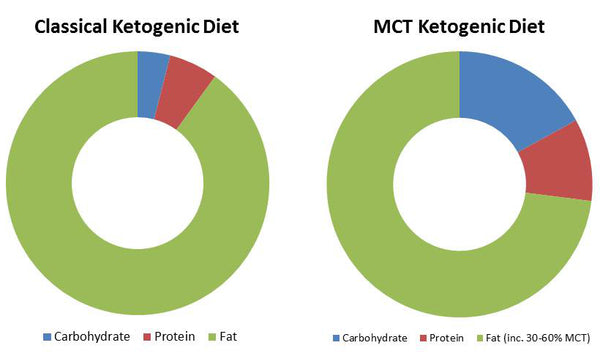
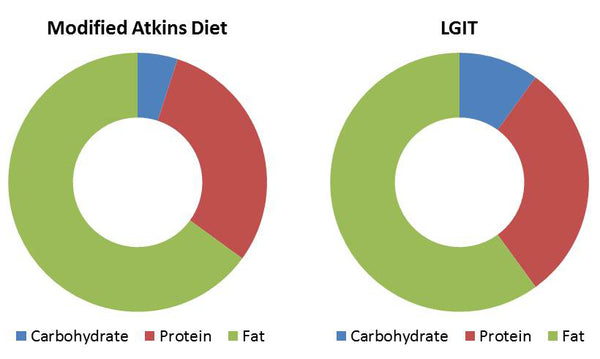
List of Clinical Trials involving Coconut - U.S. National Institute of Health
Rank NCT Number Title
1 NCT01962844 Coconut Oil in Cardiovascular Disease
2 NCT02152111 Hipoenergetic Effect of Diet Associated or Not With the Consumption of Coconut Flour in Women With Overweight
3 NCT01883648 Study to Evaluate Coconut Oil for Alzheimer's Disease
4 NCT01364948 Effect of Coconut Oil Application in Reducing Water Loss From Skin of Premature Babies in First Week of Life (TEWL)
5 NCT01758068 Effect of Twice Daily Application of Coconut Oil in Reducing Water Loss From Skin of Premature Babies in First Week of Life
6 NCT01445431 A Trial on the Efficacy and Safety of Virgin Coconut Oil Compared to Mineral Oil in the Treatment of Uremic Xerosis
7 NCT01885611 Virgin Coconut Oil Oral Supplementation for Leprosy Patients
8 NCT01821456 Cologne Cohort of Neutropenic Patients (CoCoNut)
9 NCT01876394 Comparison of Dietary Fats on Vascular Parameters
10 NCT02079571 Investigation of Pulse Waves, Channel Entries, and Food Attributes in Healthy Subjects With Different Constitutions
11 NCT02245113 Effects of High Oleic Palm Olein, Virgin Olive Oil and Coconut Oil Diets on Markers Selected in Malaysian
12 NCT01250275 Effects of Canola Oil on Blood Vessel Function in Peripheral Arterial Disease
13 NCT01381354 Nutrition, Neuromuscular Electrical Stimulation (NMES) and Secondary Progressive Multiple Sclerosis (SPMS)
14 NCT00941837 Effect of Dietary Fat Type in Combination With High Protein on Plasma Homocysteine Levels
15 NCT01378767 Health Benefits of a 21-day Daniel Fast
16 NCT01806831 Efficacy and Safety of Biocellulose Sheet Containing Anti-hyperpigmentation Agent ("Biocellulose Mask", "Farhorm®") in Patients Receiving Laser Treatment
17 NCT01396642 Topical Emollient Therapy
18 NCT01542970 Can Supplementation With Lactobacillus Reuteri and Omega-3 Fatty Acids During Pregnancy and Lactation Reduce the Risk of Allergic Disease in Infancy?
19 NCT00958685 Safety Study of Feeding With Ginger Extract in Acute Respiratory Distress Syndrome
20 NCT00396643 Indicated Prevention With Omega-3 Fatty Acids in Adolescents With 'At-Risk-Mental-State' for Psychosis
21 NCT00592072 Effect of Medium Chain Fatty Acids on Cognitive Function During Acute Hypoglycemia in Patients With Type 1 Diabetes
22 NCT01759667 DIT After RYGB - A Prospective Study
23 NCT01307748 Effects of Stress-reducing Aromatherapy
24 NCT00336908 Inflammatory Responses to Lipid Emulsions in Children Before and After Open Heart Surgery
25 NCT02142569 Effects of a Supplement Derived From Palm Oil on Cholesterol Levels and Chinese Red Yeast Rice in the Blood
26 NCT02009748 Vitamin D Supplementation in Cirrhotic Patients
27 NCT01000714 Determination of the Efficacy and Safety of Psirelax in the Relief of the Disease in Psoriasis
28 NCT01363167 Identifying Vitamin D Deficiency in Very Low Birth Weight Infant (VLBW) Infants Part 2
29 NCT02029833 Canola Oil Multi-Centre Intervention Trial II
30 NCT01595893 Vitamin D Supplementation in Polymorphic Light Eruption
31 NCT02085213 GOT-IT Trial: Glyceryl Trinitrate for Retained Placenta
32 NCT00669422 ChemoFx® PRO - A Post-Market Data Collection Study
List of Clinical Trials involving Turmeric - U.S. National Institute of Health
Rank NCT Number Title
1 NCT01906840 Role of Turmeric on Oxidative Modulation in ESRD Patients
2 NCT01015937 Effect of Turmeric on Diabetic Nephropathy
3 NCT01037595 Effect of Turmeric on Pruritus in Hemodialysis Patients
4 NCT01634256 A Human Trial to Evaluate the Efficacy and Safety of Fermented Turmeric in Subjects Showing Mild Hepatic Injury
5 NCT00992004 Evaluation of the Efficacy of a Turmeric Extract (Arantal®) in Patients With Osteoarthritis of the Knee (Gonarthrosis).
6 NCT02442453 Effect of Scaling and Root Planing Along With Topical Application of Commercially Available Curcuma Longa Gel on Superoxide Dismutase and Malondialdehyde Levels in Saliva of Chronic Periodontitis Patients
7 NCT01831193 Effect of Oral Supplementation With Curcumin (Turmeric) in Patients With Proteinuric Chronic Kidney Disease
8 NCT00969085 Trial of Curcumin in Cutaneous T-cell Lymphoma Patients
9 NCT01344291 Turmeric Effect on Reduction of Serum Prolactin and Related Hormonal Change and Adenoma Size in Prolactinoma Patients
10 NCT00219882 Safety Study of Orally Administered Curcuminoids in Adult Subjects With Cystic Fibrosis
11 NCT00792818 The Efficacy and Safety of Curcuma Domestica Extracts and Ibuprofen in Knee Osteoarthritis
12 NCT00542711 Bio-availability of a New Liquid Tumeric Extract
13 NCT02404701 Effect of Over-the-counter Dietary Supplements on Kidney Stone Risk
14 NCT02008981 Effect of Herbal Products on Platelet Function and Clotting
15 NCT00779493 Curcumin (Tumeric) in the Treatment of Irritable Bowel Syndrome: A Randomized-Controlled Trial
16 NCT01859858 A Prospective Evaluation of the Effect of Curcumin on Dose Limiting Toxicity and Pharmacokinetics of Irinotecan in Patients With Solid Tumors.
17 NCT00235625 Curcuminoids for the Treatment of Chronic Psoriasis Vulgaris
18 NCT02409381 Extract of Curcuma Longa Complexed With Phosphatidilcholine(Motore®)in the Treatment of Adults With Knee Osteoarthritis
19 NCT02300727 Study to See How Safe Curcumin is and How Well it Works When Used to Treat Mucositis in Patients Getting Chemotherapy
20 NCT01022632 Effect of Curcumin as Nutraceutical in Patients of Depression
21 NCT01418066 Ayurvedic Herbs in Diarrhea Predominant Irritable Bowel Syndrome
22 NCT02203227 Efficacy and Safety Study of Dietary Supplements in Chronic Smokers Having Mild to Moderate Hyperlipidemia
23 NCT02100202 Efficacy and Safety Study of Dietary Supplements in Chronic Smokers Having Mild to Moderate Hyperlipidemia
24 NCT01928043 Adjunctive Curcumin for Symptomatic Adolescents With Bipolar Disorder: Brain and Body Considerations
25 NCT00641147 Curcumin in Treating Patients With Familial Adenomatous Polyposis
26 NCT01917890 Radiosensitizing and Radioprotectve Effects of Curcumin in Prostate Cancer
27 NCT02172625 The Effect of Protandim Supplementation on Oxidative Damage and Athletic Performance
28 NCT01982734 Improved Oral Bioavailability of Curcumin Incorporated Into Micelles
29 NCT01066182 The DHA (Docosahexaenoic Acid) Oxford Learning and Behaviour (DOLAB) Study
30 NCT01192269 Cheek Cells - Non-invasive Fatty Acid Status Marker
31 NCT02104752 Curcumin as a Novel Treatment to Improve Cognitive Dysfunction in Schizophrenia
32 NCT01383161 18-Month Study of Curcumin
33 NCT01201694 Phase I Study of Surface-Controlled Water Soluble Curcumin (THERACURMIN CR-011L)
34 NCT00099710 Curcumin in Patients With Mild to Moderate Alzheimer's Disease
35 NCT01160302 Curcumin Biomarker Trial in Head and Neck Cancer
36 NCT01489592 Effect of Curcumin on Iron Metabolism in Healthy Volunteer
37 NCT02277223 Curcumin in Pediatric Ulcerative Colitis
38 NCT02064673 Adjuvant Curcumin to Assess Recurrence Free Survival in Patients Who Have Had a Radical Prostatectomy
39 NCT00752154 Curcumin in Rheumatoid Arthritis
40 NCT02281981 The Effects of Repeated Bouts of Downhill Running and Curcumin Supplementation on Arterial Stiffness During Recovery
41 NCT02189083 Study of Tumor-shrinking Decoction (TSD) to Treat Symptomatic Uterine Fibroids
42 NCT02213523 ABG Oxidative Stress Study Protocol-1
43 NCT01880398 Management of Fistula in Ano by Kshar Sutra, Para Surgical Procedure.
44 NCT02255370 Curcumin Associated With Thiopurin in the Prevention of Post-op Recurrence in Crohn Disease
45 NCT01514370 Dietary Supplement of Curcumin in Subjects With Active Relapsing Multiple Sclerosis Treated With Subcutaneous Interferon Beta 1a
46 NCT01294072 Study Investigating the Ability of Plant Exosomes to Deliver Curcumin to Normal and Colon Cancer Tissue
47 NCT01246973 Oral Curcumin for Radiation Dermatitis
48 NCT00977730 The Effect of Protandim on Non-alcoholic Steatohepatitis
49 NCT01811381 Curcumin and Yoga Therapy for Those at Risk for Alzheimer's Disease
50 NCT01948661 Anthocyanin Extract and Phospholipid Curcumin in Colorectal Adenoma
51 NCT01042938 Curcumin for the Prevention of Radiation-induced Dermatitis in Breast Cancer Patients
52 NCT01925287 Oral Bioavailability of Curcumin From Micronized Powder and Liquid Micelles in Healthy Young Women and Men
53 NCT00295035 Phase III Trial of Gemcitabine, Curcumin and Celebrex in Patients With Metastatic Colon Cancer
54 NCT02369549 Micro-Particle Curcumin for the Treatment of Chronic Kidney Disease
55 NCT00486460 Phase III Trial of Gemcitabine, Curcumin and Celebrex in Patients With Advance or Inoperable Pancreatic Cancer
56 NCT02213588 Effects of Plant Concentrate Blend on Oxidative Stress in Healthy Humans
57 NCT02146157 Trial of an Herb and Mineral Combination Product on Fasting Glucose in Adults at Risk for Developing Diabetes
58 NCT02476708 A Pilot Trial of Curcumin Effects on Cognition in Schizophrenia
59 NCT00954902 Effects of Antioxidants on Cardiovascular Risk Measures (Spice Study)
60 NCT01125501 Protandim and the Metabolic Syndrome
61 NCT00799630 Effects of Nutraperf Consumption in Runners
62 NCT01956500 Instaflex and Joint Pain in Community Adults
63 NCT01968564 Oral Curcumin Supplementation in Middle-Aged and Older Adults Improves Vascular Function
64 NCT01488656 Effects of Dietary Supplements on Response to Air Pollution
65 NCT02413099 The Efficacy and Safety of New Herbal Formula (KBMSI-2) in the Treatment of Erectile Dysfunction
66 NCT02321293 A Open-label Prospective Cohort Trial of Curcumin Plus Tyrosine Kinase Inhibitors (TKI) for EGFR -Mutant Advanced NSCLC
67 NCT01875822 Open-label Study of Curcumin C-3 Complex in Schizophrenia
68 NCT01333917 Curcumin Biomarkers
69 NCT01320436 Curcumin + Aminosalicylic Acid (5ASA) Versus 5ASA Alone in the Treatment of Mild to Moderate Ulcerative Colitis
70 NCT01469936 Effect of PERMEAPROTECT on the Quality of Life of Patients With Fibromyalgia
71 NCT02088307 Study of the Cardiovascular Vitamin, CardioLife
72 NCT01864473 Management of Vaginal Fistula by Kshar Sutra, a Minimally Invasive Treatment
Rank NCT Number Title
1 NCT01962844 Coconut Oil in Cardiovascular Disease
2 NCT02152111 Hipoenergetic Effect of Diet Associated or Not With the Consumption of Coconut Flour in Women With Overweight
3 NCT01883648 Study to Evaluate Coconut Oil for Alzheimer's Disease
4 NCT01364948 Effect of Coconut Oil Application in Reducing Water Loss From Skin of Premature Babies in First Week of Life (TEWL)
5 NCT01758068 Effect of Twice Daily Application of Coconut Oil in Reducing Water Loss From Skin of Premature Babies in First Week of Life
6 NCT01445431 A Trial on the Efficacy and Safety of Virgin Coconut Oil Compared to Mineral Oil in the Treatment of Uremic Xerosis
7 NCT01885611 Virgin Coconut Oil Oral Supplementation for Leprosy Patients
8 NCT01821456 Cologne Cohort of Neutropenic Patients (CoCoNut)
9 NCT01876394 Comparison of Dietary Fats on Vascular Parameters
10 NCT02079571 Investigation of Pulse Waves, Channel Entries, and Food Attributes in Healthy Subjects With Different Constitutions
11 NCT02245113 Effects of High Oleic Palm Olein, Virgin Olive Oil and Coconut Oil Diets on Markers Selected in Malaysian
12 NCT01250275 Effects of Canola Oil on Blood Vessel Function in Peripheral Arterial Disease
13 NCT01381354 Nutrition, Neuromuscular Electrical Stimulation (NMES) and Secondary Progressive Multiple Sclerosis (SPMS)
14 NCT00941837 Effect of Dietary Fat Type in Combination With High Protein on Plasma Homocysteine Levels
15 NCT01378767 Health Benefits of a 21-day Daniel Fast
16 NCT01806831 Efficacy and Safety of Biocellulose Sheet Containing Anti-hyperpigmentation Agent ("Biocellulose Mask", "Farhorm®") in Patients Receiving Laser Treatment
17 NCT01396642 Topical Emollient Therapy
18 NCT01542970 Can Supplementation With Lactobacillus Reuteri and Omega-3 Fatty Acids During Pregnancy and Lactation Reduce the Risk of Allergic Disease in Infancy?
19 NCT00958685 Safety Study of Feeding With Ginger Extract in Acute Respiratory Distress Syndrome
20 NCT00396643 Indicated Prevention With Omega-3 Fatty Acids in Adolescents With 'At-Risk-Mental-State' for Psychosis
21 NCT00592072 Effect of Medium Chain Fatty Acids on Cognitive Function During Acute Hypoglycemia in Patients With Type 1 Diabetes
22 NCT01759667 DIT After RYGB - A Prospective Study
23 NCT01307748 Effects of Stress-reducing Aromatherapy
24 NCT00336908 Inflammatory Responses to Lipid Emulsions in Children Before and After Open Heart Surgery
25 NCT02142569 Effects of a Supplement Derived From Palm Oil on Cholesterol Levels and Chinese Red Yeast Rice in the Blood
26 NCT02009748 Vitamin D Supplementation in Cirrhotic Patients
27 NCT01000714 Determination of the Efficacy and Safety of Psirelax in the Relief of the Disease in Psoriasis
28 NCT01363167 Identifying Vitamin D Deficiency in Very Low Birth Weight Infant (VLBW) Infants Part 2
29 NCT02029833 Canola Oil Multi-Centre Intervention Trial II
30 NCT01595893 Vitamin D Supplementation in Polymorphic Light Eruption
31 NCT02085213 GOT-IT Trial: Glyceryl Trinitrate for Retained Placenta
32 NCT00669422 ChemoFx® PRO - A Post-Market Data Collection Study
List of Clinical Trials involving Turmeric - U.S. National Institute of Health
Rank NCT Number Title
1 NCT01906840 Role of Turmeric on Oxidative Modulation in ESRD Patients
2 NCT01015937 Effect of Turmeric on Diabetic Nephropathy
3 NCT01037595 Effect of Turmeric on Pruritus in Hemodialysis Patients
4 NCT01634256 A Human Trial to Evaluate the Efficacy and Safety of Fermented Turmeric in Subjects Showing Mild Hepatic Injury
5 NCT00992004 Evaluation of the Efficacy of a Turmeric Extract (Arantal®) in Patients With Osteoarthritis of the Knee (Gonarthrosis).
6 NCT02442453 Effect of Scaling and Root Planing Along With Topical Application of Commercially Available Curcuma Longa Gel on Superoxide Dismutase and Malondialdehyde Levels in Saliva of Chronic Periodontitis Patients
7 NCT01831193 Effect of Oral Supplementation With Curcumin (Turmeric) in Patients With Proteinuric Chronic Kidney Disease
8 NCT00969085 Trial of Curcumin in Cutaneous T-cell Lymphoma Patients
9 NCT01344291 Turmeric Effect on Reduction of Serum Prolactin and Related Hormonal Change and Adenoma Size in Prolactinoma Patients
10 NCT00219882 Safety Study of Orally Administered Curcuminoids in Adult Subjects With Cystic Fibrosis
11 NCT00792818 The Efficacy and Safety of Curcuma Domestica Extracts and Ibuprofen in Knee Osteoarthritis
12 NCT00542711 Bio-availability of a New Liquid Tumeric Extract
13 NCT02404701 Effect of Over-the-counter Dietary Supplements on Kidney Stone Risk
14 NCT02008981 Effect of Herbal Products on Platelet Function and Clotting
15 NCT00779493 Curcumin (Tumeric) in the Treatment of Irritable Bowel Syndrome: A Randomized-Controlled Trial
16 NCT01859858 A Prospective Evaluation of the Effect of Curcumin on Dose Limiting Toxicity and Pharmacokinetics of Irinotecan in Patients With Solid Tumors.
17 NCT00235625 Curcuminoids for the Treatment of Chronic Psoriasis Vulgaris
18 NCT02409381 Extract of Curcuma Longa Complexed With Phosphatidilcholine(Motore®)in the Treatment of Adults With Knee Osteoarthritis
19 NCT02300727 Study to See How Safe Curcumin is and How Well it Works When Used to Treat Mucositis in Patients Getting Chemotherapy
20 NCT01022632 Effect of Curcumin as Nutraceutical in Patients of Depression
21 NCT01418066 Ayurvedic Herbs in Diarrhea Predominant Irritable Bowel Syndrome
22 NCT02203227 Efficacy and Safety Study of Dietary Supplements in Chronic Smokers Having Mild to Moderate Hyperlipidemia
23 NCT02100202 Efficacy and Safety Study of Dietary Supplements in Chronic Smokers Having Mild to Moderate Hyperlipidemia
24 NCT01928043 Adjunctive Curcumin for Symptomatic Adolescents With Bipolar Disorder: Brain and Body Considerations
25 NCT00641147 Curcumin in Treating Patients With Familial Adenomatous Polyposis
26 NCT01917890 Radiosensitizing and Radioprotectve Effects of Curcumin in Prostate Cancer
27 NCT02172625 The Effect of Protandim Supplementation on Oxidative Damage and Athletic Performance
28 NCT01982734 Improved Oral Bioavailability of Curcumin Incorporated Into Micelles
29 NCT01066182 The DHA (Docosahexaenoic Acid) Oxford Learning and Behaviour (DOLAB) Study
30 NCT01192269 Cheek Cells - Non-invasive Fatty Acid Status Marker
31 NCT02104752 Curcumin as a Novel Treatment to Improve Cognitive Dysfunction in Schizophrenia
32 NCT01383161 18-Month Study of Curcumin
33 NCT01201694 Phase I Study of Surface-Controlled Water Soluble Curcumin (THERACURMIN CR-011L)
34 NCT00099710 Curcumin in Patients With Mild to Moderate Alzheimer's Disease
35 NCT01160302 Curcumin Biomarker Trial in Head and Neck Cancer
36 NCT01489592 Effect of Curcumin on Iron Metabolism in Healthy Volunteer
37 NCT02277223 Curcumin in Pediatric Ulcerative Colitis
38 NCT02064673 Adjuvant Curcumin to Assess Recurrence Free Survival in Patients Who Have Had a Radical Prostatectomy
39 NCT00752154 Curcumin in Rheumatoid Arthritis
40 NCT02281981 The Effects of Repeated Bouts of Downhill Running and Curcumin Supplementation on Arterial Stiffness During Recovery
41 NCT02189083 Study of Tumor-shrinking Decoction (TSD) to Treat Symptomatic Uterine Fibroids
42 NCT02213523 ABG Oxidative Stress Study Protocol-1
43 NCT01880398 Management of Fistula in Ano by Kshar Sutra, Para Surgical Procedure.
44 NCT02255370 Curcumin Associated With Thiopurin in the Prevention of Post-op Recurrence in Crohn Disease
45 NCT01514370 Dietary Supplement of Curcumin in Subjects With Active Relapsing Multiple Sclerosis Treated With Subcutaneous Interferon Beta 1a
46 NCT01294072 Study Investigating the Ability of Plant Exosomes to Deliver Curcumin to Normal and Colon Cancer Tissue
47 NCT01246973 Oral Curcumin for Radiation Dermatitis
48 NCT00977730 The Effect of Protandim on Non-alcoholic Steatohepatitis
49 NCT01811381 Curcumin and Yoga Therapy for Those at Risk for Alzheimer's Disease
50 NCT01948661 Anthocyanin Extract and Phospholipid Curcumin in Colorectal Adenoma
51 NCT01042938 Curcumin for the Prevention of Radiation-induced Dermatitis in Breast Cancer Patients
52 NCT01925287 Oral Bioavailability of Curcumin From Micronized Powder and Liquid Micelles in Healthy Young Women and Men
53 NCT00295035 Phase III Trial of Gemcitabine, Curcumin and Celebrex in Patients With Metastatic Colon Cancer
54 NCT02369549 Micro-Particle Curcumin for the Treatment of Chronic Kidney Disease
55 NCT00486460 Phase III Trial of Gemcitabine, Curcumin and Celebrex in Patients With Advance or Inoperable Pancreatic Cancer
56 NCT02213588 Effects of Plant Concentrate Blend on Oxidative Stress in Healthy Humans
57 NCT02146157 Trial of an Herb and Mineral Combination Product on Fasting Glucose in Adults at Risk for Developing Diabetes
58 NCT02476708 A Pilot Trial of Curcumin Effects on Cognition in Schizophrenia
59 NCT00954902 Effects of Antioxidants on Cardiovascular Risk Measures (Spice Study)
60 NCT01125501 Protandim and the Metabolic Syndrome
61 NCT00799630 Effects of Nutraperf Consumption in Runners
62 NCT01956500 Instaflex and Joint Pain in Community Adults
63 NCT01968564 Oral Curcumin Supplementation in Middle-Aged and Older Adults Improves Vascular Function
64 NCT01488656 Effects of Dietary Supplements on Response to Air Pollution
65 NCT02413099 The Efficacy and Safety of New Herbal Formula (KBMSI-2) in the Treatment of Erectile Dysfunction
66 NCT02321293 A Open-label Prospective Cohort Trial of Curcumin Plus Tyrosine Kinase Inhibitors (TKI) for EGFR -Mutant Advanced NSCLC
67 NCT01875822 Open-label Study of Curcumin C-3 Complex in Schizophrenia
68 NCT01333917 Curcumin Biomarkers
69 NCT01320436 Curcumin + Aminosalicylic Acid (5ASA) Versus 5ASA Alone in the Treatment of Mild to Moderate Ulcerative Colitis
70 NCT01469936 Effect of PERMEAPROTECT on the Quality of Life of Patients With Fibromyalgia
71 NCT02088307 Study of the Cardiovascular Vitamin, CardioLife
72 NCT01864473 Management of Vaginal Fistula by Kshar Sutra, a Minimally Invasive Treatment
Last edited: